SERVICE
End-End CQV Planning
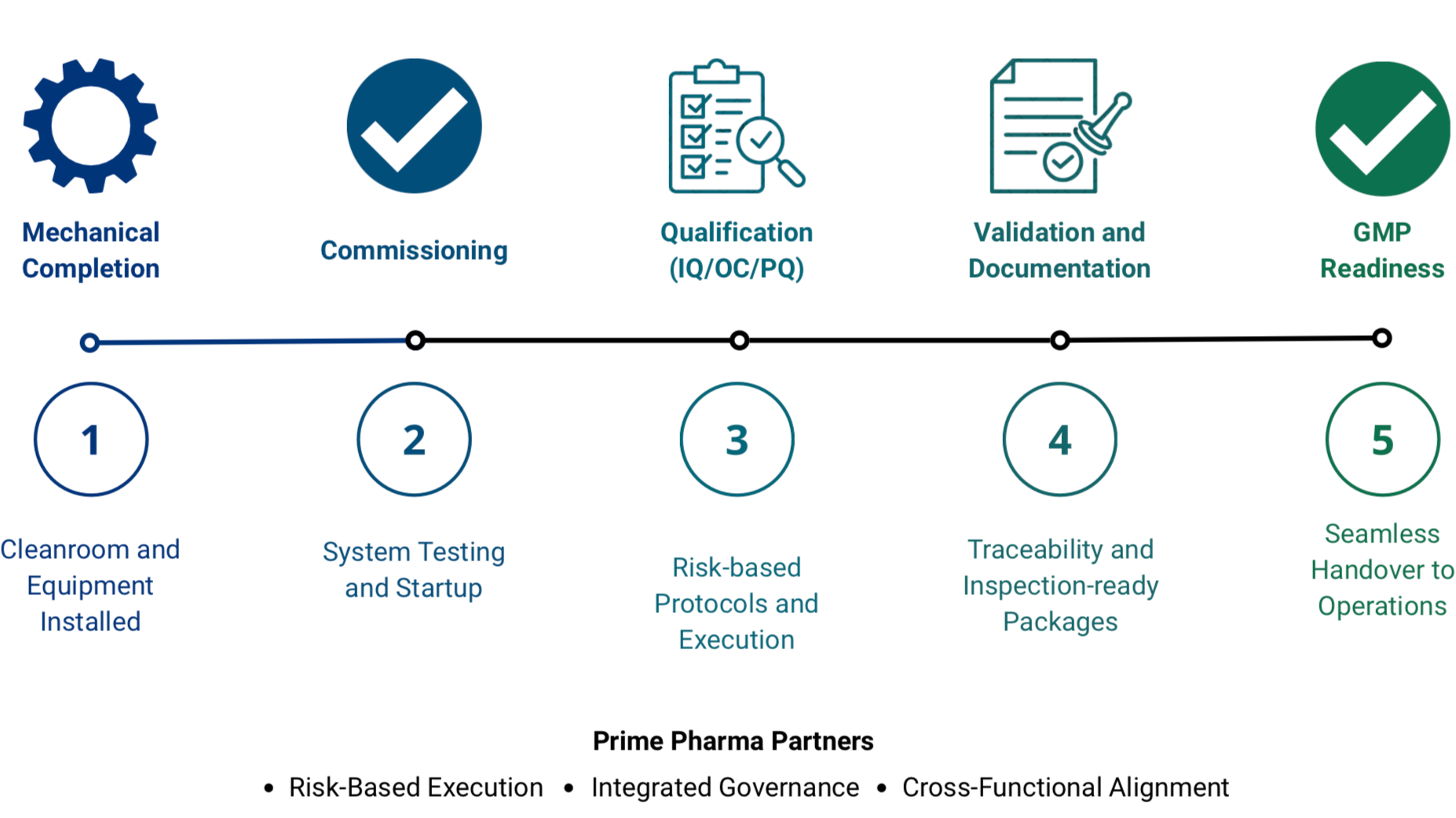
From Construction Completion → GMP Readiness
We deliver integrated governance, risk-based execution, and true cross-functional alignment so you avoid late-stage surprises, stay on schedule, and reach GMP-ready commercial manufacturing faster and with greater confidence.
What We Do
Prime Pharma Partners leads CQV programs from construction completion through GMP readiness, integrating engineering, validation, automation, and quality into a single execution framework. We establish governance, control dependencies, and align cross-functional teams to ensure predictable, inspection-ready delivery.
❋ Where This Fits
Post-construction / mechanical completion
Inspection readiness and regulatory preparation
CQV execution and qualification phases
Pre-GMP readiness and startup
What We Provide
-

CQV program leadership and execution ownership.
-

Integrated master scheduling (IMS) and dependency control.
-

Cross-functional alignment across Engineering, Validation, QA, Automation, and Manufacturing.
-
Governance structures and escalation pathways to maintain milestone integrity.
-
Strategy development for qualification, readiness, and closeout.
What Prime Pharma Partners Provides
We establish integrated program governance and scheduling frameworks that provide full visibility into execution, enabling leadership to make informed decisions, resolve constraints, and maintain control of GMP readiness milestones.
CQV program leadership and execution ownership.
Integrated master scheduling (IMS) and dependency control.
Cross-functional alignment across Engineering, Validation, QA, Automation, and Manufacturing.
Governance structures and escalation pathways to maintain milestone integrity.
Strategy development for qualification, readiness, and closeout.
❋ Common Deliverables
End-to-end CQV execution plans and integrated schedules.
Qualification protocols (IQ/OQ/PQ) aligned to execution strategy.
Risk registers, issue logs, and readiness tracking tools.
System turnover and closeout documentation.
Inspection-ready validation and GMP evidence packages.
What Prime Pharma Partners Provides
-
CQV program leadership and execution ownership.
It all begins with an idea. Maybe you want to launch a business. Maybe you want to turn a hobby into something more. Or maybe you have a creative project to share with the world. Whatever it is, the way you tell your story online can make all the difference.
-
Build it.
It all begins with an idea. Maybe you want to launch a business. Maybe you want to turn a hobby into something more. Or maybe you have a creative project to share with the world. Whatever it is, the way you tell your story online can make all the difference.
-
Grow it.
It all begins with an idea. Maybe you want to launch a business. Maybe you want to turn a hobby into something more. Or maybe you have a creative project to share with the world. Whatever it is, the way you tell your story online can make all the difference.
What We Do
Prime Pharma Partners leads CQV programs from construction completion through GMP readiness, integrating engineering, validation, automation, and quality into a single execution framework. We establish governance, control dependencies, and align cross-functional teams to ensure predictable, inspection-ready delivery.
❋ Where This Fits
Controlled, schedule-driven execution aligned to GMP milestones.
Reduced late-stage risk through proactive dependency management.
Inspection-ready systems and documentation.
Clear ownership and accountability across all workstreams.
❋ What Prime Pharma Partners Provides
We establish integrated program governance and scheduling frameworks that provide full visibility into execution, enabling leadership to make informed decisions, resolve constraints, and maintain control of GMP readiness milestones.
CQV program leadership and execution ownership.
Integrated master scheduling (IMS) and dependency control.
Cross-functional alignment across Engineering, Validation, QA, Automation, and Manufacturing.
Governance structures and escalation pathways to maintain milestone integrity.
Strategy development for qualification, readiness, and closeout.
❋ Common Deliverables
End-to-end CQV execution plans and integrated schedules.
Qualification protocols (IQ/OQ/PQ) aligned to execution strategy.
Risk registers, issue logs, and readiness tracking tools.
System turnover and closeout documentation.
Inspection-ready validation and GMP evidence packages.
❋ Outcomes
Post-construction / mechanical completion
CQV execution and qualification phases
Pre-GMP readiness and startup
Inspection readiness and regulatory preparation
End-to-End CQV: From Construction Completion to GMP-Ready